E-Archive
Science Update
in Vol. 15 - September Issue - Year 2014
Investigating the effects of laser shock peening without absorbent coating (LSPwC) on residual stresses and corrosion behavior of AA6082

Fig. 1: a) Experimental LSPwC set-up; b) generation of confined plasma during LSPwC and c) schematic presentation of the LSPwC surface scan/sweep
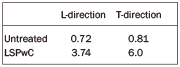
Table 1. Surface roughness (Ra) results.
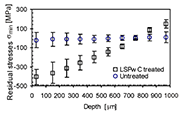
Fig. 2. Hole drilling principal residual stresses -
![Fig 3. Open circuit potential (Eocp = f [t]) curves in 35 wt.% NaCl solution](https://mfn.li/storage/e-archives/article-pictures/1419/3936.gif)
Fig 3. Open circuit potential (Eocp = f [t]) curves in 35 wt.% NaCl solution
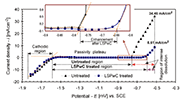
Fig 4. Linear voltammograms of the untreated and LSPwC treated specimen
Introduction
Aluminium alloys are frequently used in industrial applications due to their low density, favourable mechanical properties, and high corrosion resistance. However, despite the general high corrosion resistance of Al alloys in a chloride environment, localized corrosion and stress corrosion cracking often occur [1]. Therefore, an adequate surface treatment is essential to increase corrosion, fatigue, and wear resistance [2-4]. Laser Shock Peening (LSP) is an innovative surface treatment that is mostly applied to improve fatigue strength and fatigue crack initiation life through the creation of compressive residual stresses in the surface layer. When processing the material surface with laser pulse with appropriate characteristics, the material¡¦s surface is vaporized and reaches temperatures of 104 ¢XC or more, which produce plasma. Plasma confined in water produces shock impact waves, which, in turn, produce considerable densification of dislocations and generation of compressive residual stresses of high gradient. In practice, LSP has already been shown to produce higher compressive residual stresses to much greater depths comparable to conventional shot peening (SP). In addition, after the SP process, the surface of the metal component can be damaged and distorted, while after LSP, the surface of the treated component is essentially unaffected. The objective of the present study was to investigate the effects of LSP on the enhancement of surface integrity and corrosion resistance of peak-aged AA6082-T651 aluminium alloy.
Experimental
The material tested was a 6082-T651 aluminium alloy supplied as a drawn round bar of Ø40 mm with composition in wt. % as Si: 0.98, Mg: 0.67, Mn: 0.53, Fe: 0.25, Ti: 0.03, some minor elements (Cr, Zn, Ti, Cu) and the remaining was Al. Static mechanical properties of the tested material were: Rm=348 MPa, Rp0.2=320 MPa and HV=92.
LSPwC treatment (Fig. 1) was performed in the Centro Laser U.P.M. Madrid, Spain using the method of closed ablation without protective coating (LSPwC), with a Q-switched Nd:YAG laser of 1064 nm. Power density of 10.75 GW/cm2, with pulse duration (FWHM) of 10 ns and repetition rate of 10 Hz was used. A pulse density of 900 pulses/cm2 was equivalent to a final cumulative fluence of 1.71 kJ/cm2. A schematic presentation of the LSPwC process is given in Fig. 1. Residual stresses (RS) prior to and after LSPwC treatment were measured with a semi destructive hole-drilling method in accordance with ASTM E 837, where the final RS was determined by the integral incremental method. Corrosion tests were conducted in 3.5 wt. % NaCl water (pH 7, 23 ¢XC). The electrochemical characteristics were evaluated using a potentiostatic Voltalab 21, Radiometer Analytical and three-electrode CEC/TH cell with a Pt counter and a reference calomel electrode (SCE = +244 mV vs. SHE). In order to stabilize the surface, the specimens were immersed in the medium for 60 min during which the open-circuit potential (OCP) was monitored. Afterwards, linear voltammetry test were executed. In order to observe cathodic corrosion phenomena at lower potentials, the measurements started at ¡V1800 mVSCE, and were shifted up to ¡V500 mVSCE at 10 mVs-1.
Results and discussion
Surface roughness
Due to the preliminary preparation of the specimens with a cutter and the LSPwC surface sweep direction, it was decided to establish surface profiles in the longitudinal (L) and transverse (T) directions by mean values of the arithmetic roughness and
using three profile measurements (Table 1).
Roughness Ra of untreated specimen was 0.72 £gm in the the L direction and 0.81 £gm in the T direction. In the LSPwC treated specimen, Ra was to 3.74 £gm in L direction and 6.0 £gm in T direction. The increase in surface roughness after LSPwC is rather logical, due to the numerous laser-beam interactions with the specimen surface due to pulse overlapping and the cumulative action of induced plasma and shock waves at the interaction point.
Residual stresses
The principal residual stresses (RS) as a function of depth are shown in Fig. 2. The principal residual stresses £mmax and £mmin represent the stress component in the longitudinal (L) and transverse (T) of the LSPwC-scan directions respectively.
The residual stresses in the initial state specimen were about the same in both longitudinal and transverse directions, and they ranged from +47¡Ó 15 MPa to −21¡Ó7 MPa. Analysis of the principal RS after LSPwC treatment has shown a predominantly compressive character. It was observed that in the case of LSPwC treatment, £mmax and £mmin changed to the tensile state after reaching depths of approximately 620 £gm and 770 £gm, respectively. Thus, higher hardening depth (higher compression) due to shock waves was obtained with minimal principal stresses, i.e. in the T direction of the LSPwC sweep direction. Furthermore, the highest compressive RS at the near surface (33 £gm) after LSPwC was obtained in the transverse direction £mmin =−407¡Ó81 MPa. Afterwards, a constant decrease with a constant gradient was observed up to a final depth of 967 £gm. Due to excessive shock wave pressure at the specimen¡¦s surface, the highest compressive RS exceeded the "quasi" static tensile strength (Rm = 327 MPa) of the base material.
Corrosion analysis
From the OCP transients (Eocp=f[t]) in Fig. 3, it is obvious that the untreated specimen exhibits more negative values compared to the LSPwC specimen. Moreover, relatively stable and higher potential values are obtained after LSPwC, indicating an equilibrium state at which the rate of oxidation (IOx) equals that of reduction (IRed). Such behaviour after LSPwC treatment is most likely attributed to the formation of a modified aluminium oxide and nano-structure in the treated area that enhances the material¡¦s corrosion resistance and mechanical properties.
Results of linear voltammograms for both untreated and LSPwC treated specimens (Fig. 4) confirmed a relatively high cathodic current of ¡V 14 mA/cm2 at the stating potential of -1.8 VSCE. Afterwards, a sharp decrease occurred due to formation of Al-oxide at the specimen¡¦s surface at the potential near ¡V 1.6 VSCE where the current density was practically zero, which is characteristic of a passivity plateau. The current density in the anodic scan in the passive region stayed practically unchanged up to ¡V 0.782 VSCE for the untreated specimen. However, in the case of the LSPwC treated specimen, the region of passivity greatly increased, i.e. up to a potential of ¡V 0.64 VSCE. The rapid increase in the subsequent anodic potential represents the transpassive region, which is directly associated with the dissolution of Al alloy. Apparently, despite increased roughness, the LSPwC treated specimen exhibited an improved and extended region of passivity, indicating improved corrosion resistance compared to the untreated specimen. Furthermore, the anodic current at ending potential (¡V0.5 VSCE) confirmed a 4¡Vfold decrease after LSPwC treatment (34.46 mA/cm2 vs. 8.61 mA/cm2).
One should note that in the present study, LSP was performed without a protective overlay (LSPwC) on the surface of the processed specimens. Thus, heat generated from the laser pulses affect the surface state due to surface melting and plasma ionisation indicating a chemically stable phase after LSPwC, which protected the material against corrosion attack.
As can be seen in Fig. 5, the localized pitting attack is present in both specimens; nevertheless, the intensity of pitting attack with a presence of large corrosion product (CP) is more severe in the untreated one, with intense, wide-spread pitting attack.
The pit shape in the untreated sample was mostly "semi" circular or "semi" elliptical), with diameters in the 100¡V300 £gm range. However, after LSPwC treatment, significant modifications are obtained, with only a few smaller hemi-spherical anodic pits observed.
The cathodic corrosion and the accumulation of OH¡Â ions occurs at potentials more negative than - 1130 mVSCE, with pitting attack presented in the form of bright surface craters
From all of the above-mentioned, it is clear that LSPwC treatment clearly makes the penetration of water molecules, oxygen and Cl¡Â ions more difficult due to the development of compressive residual stresses in the near surface and an improved passive/oxide film after LSPwC treatment [5][6]. In fact, more positive corrosion potential, and an extended region of passivity with reduced anodic current, indicate a lower advance of corrosion in the LSPwC specimen in an aggressive chloride environment.
Conclusion
LSPwC surface treatment has turned out to be a technique sufficient to induce strain hardening and establish high compressive residual stresses of -242 MPa in the surface. However, surface roughness after LSPwC increased as a consequence of numerous laser ablation, plasma and shock waves on the treated surface. OCP transients confirmed more positive and stable potential values of the LSPwC treated specimen, indicating improved corrosion resistance. In addition, voltammetry tests confirmed the improved characteristics after LSPwC, showing a larger region of passivity, a smaller transpassive region, and exceptionally reduced anodic current density compared to the untreated specimen. Localized pitting corrosion attack was observed in both the untreated and LSPwC treated state. However, despite the increased roughness of the LSPwC specimen, the extent of passivity occurring and localized pitting attack was reduced, which confirmed improved corrosion resistance. Improved resistance to localized corrosion is attributed to the transformed oxide film created due to ablation of the specimen surface during the laser peening process and high compressive residual stresses, both of which hinder corrosion progress.
References ¡V Further reading
[1] Z. Szklarska-Smialowska. Corros. Sci. 41 (1999) 1742-1764.
[2] U. Trdan, J.L. Ocaña, J. Grum. J. Mech. Eng. 57(5) (2011) 385-393.
[3] U. Sánchez-Santana, C. Rubio-González, G. Gomez-Rosas, J.L. Ocaña, C. Molpeceres, J.A. Porro J, M. Morales M. Wear 260 (2006) 847-854.
[4] U. Trdan, J.A. Porro, J.L. Ocaña, J. Surf. Coat. Technol. 208 (2012) 109-116.
[5] U. Trdan, J. Grum J. Corros. Sci. 82 (2014) 328-338.
[6] U. Trdan, J. Grum. Corros. Sci. 59 (2012) 324-333.
For Information:
U. Trdana; J.A. Porrob; J.L. Ocañab; J. Gruma
aUniversity of Ljubljana
Faculty of Mechanical Engineering, Slovenia
bCentro Láser U.P.M.
Carretera de Valencia km. 7,300
28031 Madrid, Spain





























